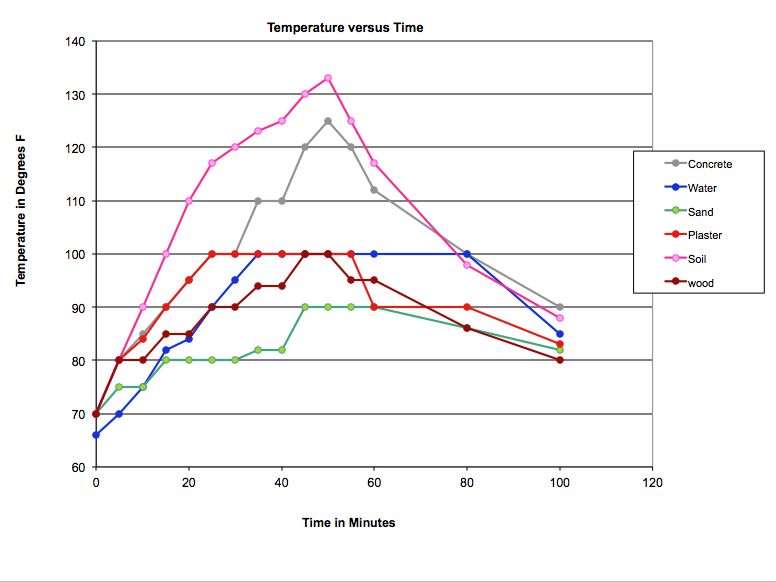
We conducted a number of experiments on passive solar heating, including one which compared how rapidly various building materials absorbed heat from the sun, and how quickly they lost it when the "sun" went down.
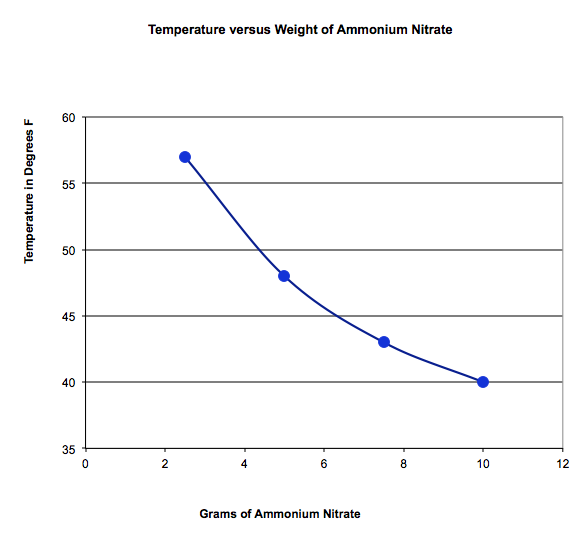
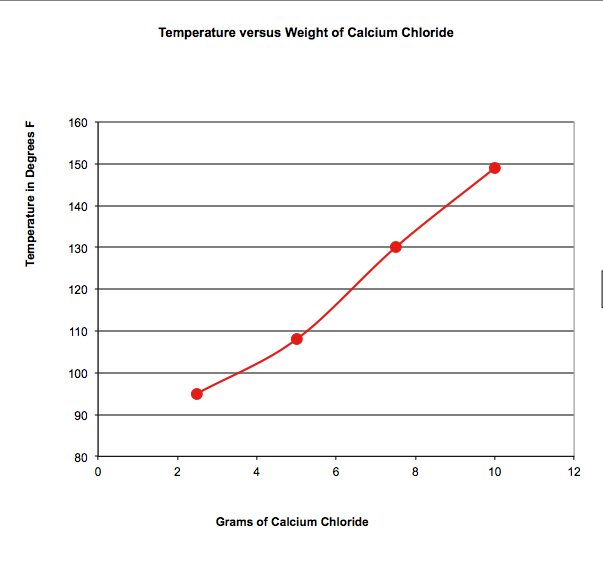
We investigated an endothermic reaction, one that gets cold, by mixing ammonium nitrate from an instant cold pack with water, and an exothermic reaction, one that gets hot, by mixing calcium chloride ice melt with water. How did the amount of material effect the temperature of the solutions?
The Wednesday group designed insulated containers to keep a block of ice from melting. Teams chose from newspaper, sawdust, foam sheets, polystyrene beads, packing peanuts, aluminum foil, and bubble wrap, and a assortment of various jars, canisters, and bottles. None of them kept the ice from melting after eight hours so we repeated the experiment for only 2.5 hours. The most successful device retained 73% of the original ice by weight, built by Varun J. and Nathan H..
We also explored the effectiveness of various types of fabrics and layering on keeping warm. Students selected a base layer (underwear), middle layer (outfit), and outer layer (coat) to dress an aluminum cylinder "student". We filled the dressed "students" with very hot water, added a thermometer, and monitored their tempertatures throughout the rest of the meeting. All of Tuesday's "students" stayed pretty warm indoors (shown below), retaining about 70% of their heat. "Alexa", dressed by Michelle, Alexandra K., Sophia H., and Kanon in a cotton jersey undershirt, velvet dress, and lined vinyl raincoat, did best at 75%. We put Wednesday's "students" outside on what was a cold afternoon. "Steve Bob Skittle III", dressed by Owen, Eric, Emily, and Freddy in a cotton jersey undershirt, polyester fleece shirt, and fur coat lost only 20 degrees, retaining 89% of its heat.